The Federal Ministry of Education and Research (BMBF) has been funding research in the field of zoonoses since 2008.
Since 2017, the Research Network for Zoonotic Infectious Diseases has been in existence, which is located at many different research institutes throughout Germany. The Institute of Virology at Charité - Universitätsmedizin Berlin is in charge of the coordination project of the research network.
Under the umbrella of the Research Network Zoonotic Infectious Diseases projects of the Public Health Service (ÖGD) and the Veterinary Service are funded.
Background
Zoonotic infections - i.e. infectious diseases that can be mutually transmitted between humans and animals - are a constant challenge for society. They are of particular concern to the scientific community in the fields of human and veterinary medicine, affect the population in the form of individual diseases and place particular demands on the Public Health Service (ÖGD) and the veterinary service. Interdisciplinary work is a prerequisite for effectively combating zoonoses.
Funding
In order to support the research, detection, prevention and cure of zoonoses in the operative field, the Federal Ministry of Education and Research (BMBF) funds so-called ÖGD projects under the umbrella of the Research Network for Zoonotic Infectious Diseases. These projects are intended to enable institutions of the public health service and the veterinary service to scientifically investigate concrete challenges in their region and in their everyday life in the field of zoonoses. For this purpose, they can receive BMBF funding upon application. If required, members of the research network can provide support in the development of research questions and in research.
Applications can be submitted in response to the respective calls for proposals. Calls were launched in 2018 and 2019, but currently there is no call and no possibility of submission.
ÖGD-Projects
Research projects of the Public Health Service (ÖGD) - so-called ÖGD projects - are dedicated to concrete topics proposed by institutions of the ÖGD and the veterinary sector. The topics are varied and are described in the respective project descriptions.
Current ÖGD projects
Many applicants followed the call for ÖGD projects in 2018. Numerous submitted projects were recommended for funding by the coordination committee. The following projects are already being funded:
Grant recipients: Dr. Stefanie Heinze / Prof. Dr. Caroline Herr
Institution: Bayerisches Landesamt für Gesundheit und Lebensmittelsicherheit
Funding period: 1.1.2019 - 31.12.2021
Brief description
For some years now, the number of infections caused by multi-resistant pathogens (MRE) has been increasing.
The most important pathogens with antibiotic resistance are multi-resistant Gram-negative
pathogens (MRGN) such as extended-spectrum β-lactamase-producing enterobacteria (ESBL) and methicillin-resistant Staphylococcus aureus (MRSA). The aspect of zoonotic significance of MRE is also playing an increasingly important role in the private sector, but is not addressed much in previous recommendations. In addition, many people (farmers, veterinarians and their household members) acquire MRE through contact with farm animals.
The publications of the Commission for Hospital Hygiene and Infection Prevention (KRINKO) at the Robert Koch Institute or the Technical Rules for Biological Agents (TRBA) focus on the formulation of recommendations for health care facilities for the prevention of nosocomial infections or prevention of occupational infection risks. Recommendations for the private sector or for non-medical institutions are missing. The topic of the zoonotic importance of antibiotic resistance is touched upon in the KRINKO's comments, but no specific advice is given for this area; this is also missing in the TRBAs (250 for health care workers; 230 for agriculture/forestry; TRBA 260 veterinary medicine comparable activities). As a consequence, the Public Health Service (PHS) lacks answers to many questions concerning MRE in the private sector, including zoonotic MRE.
The ZooM project aims to identify and answer the open questions relevant to the work of the ÖGD (human and veterinary medicine) in the field of zoonotic significance of MRE at the veterinary/human medicine interface and to disseminate them as FAQs. Subsequently, the FAQs and their dissemination will be evaluated with regard to their suitability for the daily work of the ÖGD (human and veterinary medicine) and their dissemination.
The project ZooM is funded by the Federal Ministry of Education and Research (BMBF) within the framework of the Research Network Zoonotic Infectious Diseases.
Beneficiary: Dr. Sylvia Klees
Institution: Chemical and Veterinary Investigation Office Ostwestfalen-Lippe (CVUA-OWL)
Funding period: 1.10.2018 - 30.6.2020
Brief description
The increasing spread of multi-resistant pathogens (MRE) is one of the most important threats to human and animal health. An increase in resistance to antibiotics leads to the ineffectiveness of drugs, so that an infectious disease can no longer be treated or cannot be treated sufficiently. Monitoring the emergence of antimicrobial resistance in humans and food-producing animals and identifying the molecular mechanisms of resistance transmission are important aspects for developing effective strategies to control and prevent antimicrobial resistance. MRE occur in different animal species, the environment, feed, food and humans and can be transmitted from animals to humans via the food derived from animals, such as meat. (BfR Homepage)
Zoonotic transmission has already been demonstrated for several MRE (e.g. methicillin-resistant Staphylococcus aureus (MRSA), extended spectrum β-lactamase (ESBL)-forming enterobacteria (ESBL-E)), with direct transmission between farm animal and exposed humans confirmed as a major transmission pathway.
Besides MRSA and ESBL-E, zoonotic transmission pathways have recently been considered for a number of other MRE. [1].
- Carbapenems are beta-lactam antibiotics that are only used in human medicine and belong to the group of "highest priority critically important antimicrobials". Nevertheless, carbapenemase-forming enterobacteria (CRE) have been detected in livestock farms in Germany as well.
- Recently, the spread of plasmid-encoded colistin resistance genes (mcr) in meat samples, farm animals and humans has been observed in China and in several European countries including Germany [2]. Colistin is used in human medicine as a reserve antibiotic when other antibiotics are no longer effective, so this development threatens the success of therapeutic measures in humans. In veterinary medicine, colistin is used in Germany at a rate of about 107 t per year (in 2014).
- After the ban of Avoparcin as a growth promoter in the 1990s, the proportion of vancomycin-resistant enterococci (AER) in meat samples decreased, but some recent studies still found a high prevalence in pigs in Portugal (25%) and a low prevalence in biogas plants in Germany [3,4]. In addition, novel transferable resistance genes have been found in China in enterococci from animal holdings and hospitals, which also mediate insensitivity to human reserve antibiotics (oxazolidinones) (optrA) [5].
The aim of our study is to investigate the occurrence of defined MRE or resistance determinants (ESBL, CRE/carbapenemases, colistin, optrA, AER) beyond the routine investigations performed in the framework of EU-wide monitoring programmes in the (poultry) meat production and processing chain as well as in environmental samples (e.g. water samples from lakes and springs) by means of cultural and molecular biological methods. The aim is to identify sources of input and investigate possible further spread in the meat processing chain and into the environment.
A further characterisation (whole genome typing) of the isolated strains will be carried out in cooperation with the consortium #1Health-PREVENT by the project partner University Hospital Münster. This study is intended to complement the investigations of the nationwide zoonoses monitoring in 2018 and 2019 and will run for two years.
The results of the project are to be used to identify sources of entry and contamination pathways through a fine typing of resistant strains. The occurrence of MRE (ESBL/CRE/Colistin resistance/VRE) in meat processing will be examined and sources of contamination identified in order to better assess the risk potential of consumers through contact with food of animal origin. The results should also give reason to check the hygiene management (especially cleaning and disinfection) of the establishments for weak points.
This study is part of the National Research Network on Zoonotic Infectious Diseases and is funded by the Federal Ministry of Education and Research (BMBF) under the funding code 01KI1807.
1. Carbapenem-resistant Enterobacteriaceae in wildlife, food-producing, and companion animals: a systematic review. Köck R, Daniels-Haardt I, Becker K, Mellmann A, Friedrich AW, Mevius D, Schwarz S, Jurke A. Clin Microbiol Infect. 2018 Apr 11. pii: S1198-743X(18)30339-2. doi: 10.1016/j.cmi.2018.04.004. [Epub ahead of print]
2. Prevalence of mcr-1 in E. coli from Livestock and Food in Germany, 2010-2015. Irrgang A, Roschanski N, Tenhagen BA, Grobbel M, Skladnikiewicz-Ziemer T, Thomas K, Roesler U, Käsbohrer A. PLoS One. 2016 Jul 25;11(7):e0159863. doi: 10.1371/journal.pone.0159863.
3. Genetic characterisation of antibiotic resistance and virulence factors in vanA-containing enterococci from cattle, sheep and pigs subsequent to the discontinuation of the use of avoparcin. Ramos S, Igrejas G, Rodrigues J, Capelo-Martinez JL, Poeta P. Vet J. 2012 Jul;193(1):301-3. doi: 10.1016/j.tvjl.2011.12.007.
4. Cultivation of vancomycin-resistant enterococci and methicillin-resistant staphylococci from input and output samples of German biogas plants. Glaeser SP, Sowinsky O, Brunner JS, Dott W, Kämpfer P. FEMS Microbiol Ecol. 2016 Mar;92(3). pii: fiw010. doi: 10.1093/femsec/fiw010.
5. A novel gene, optrA, that confers transferable resistance to oxazolidinones and phenicols and its presence in Enterococcus faecalis and Enterococcus faecium of human and animal origin. Wang Y, Lv Y, Cai J, Schwarz S, Cui L, Hu Z, Zhang R, Li J, Zhao Q, He T, Wang D, Wang Z, Shen Y, Li Y, Feßler AT, Wu C, Yu H, Deng X, Xia X, Shen J. J Antimicrob Chemother. 2015 Aug;70(8):2182-90. doi: 10.1093/jac/dkv116.
Beneficiary of the grant: Dr. Ute Messelhäußer
Institution: Bavarian State Office for Health and Food Safety
Funding period: 1.1.2019 - 31.12.2021

Fig. 1: Egg huts - an increasingly frequent point of sale for table eggs
(Source: District Office Weilheim-Schongau - Jens Lewitzki)
Thermophilic Campylobacter spp. is currently one of the most important food-borne zoonotic agents. Besides ruminants, the main reservoir of this pathogen is (domestic) poultry. It is currently assumed that the majority of human cases of the disease are due to the consumption of not fully cooked poultry meat or cross-contamination when using raw poultry meat in the household. To what extent also table eggs (class A eggs) play a role in the transmission of the pathogen to humans (transmission via consumption of raw poultry meat or cross-contamination) is still little known. In order to characterise the risk of contamination of eggs of class A with thermophilic Campylobacter spp. in more detail and to be able to develop appropriate reduction strategies if there are indications of relevance of eggs with regard to transmission of thermophilic Campylobacter spp. to humans, the following questions will be addressed within the framework of the project:
- What factors determine the survival of thermophilic Campylobacter spp. on eggs
- To what extent is there also the possibility of survival of thermophilic Campylobacter spp. in eggs, for which only few, partly contradictory study results exist so far?
- To what extent can a possible correlation between the consumption of eggs or the handling of raw eggs and human Campylobacteriosis be established in epidemiological as well as molecular biological studies?
- Which possibilities for reducing the introduction of thermophilic Campylobacter spp. via the food chain "egg" can be implemented in practice, especially in farming systems?
Work package 2 will investigate the correlation between egg consumption and the handling of raw eggs and human campylobacterioses
(epidemiology). This is done on the one hand by interviewing patients with human cases of campylobacteriosis using a standardised questionnaire on the respective consumption habits with special consideration of eggs and raw food (classical epidemiology) and on the other hand by obtaining human
Campylobacter isolates (symptomless eliminators and cases of disease) and the comparison of human, veterinary and food-associated Campylobacter isolates using molecular biological methods, e.g. PFGE, MLST, NGS (Molecular Epidemiology).
In work package 3, possible reduction measures for the introduction of thermophilic Campylobacter spp. via the food chain "egg" will be worked out, taking into account the practical feasibility, especially in farming systems.
Utilization of results:
It is planned to use and publish the results within the framework of two dissertation projects (including presentation of the results at conferences and publication in peer-reviewed journals). Furthermore, the results, in particular the development of reduction measures on farms, should result in guidelines for both agriculture and official food control, so that possible reduction measures can also be implemented in the area within the framework of primary production.
Cooperation partners:
- Bayerisches Landesamt für Gesundheit und Lebensmittelsicherheit (LGL)
- Abteilung Landesinstitut Planung und Lenkung Lebensmittelsicherheit, Sachbereich Fachgruppe Zoonosen, Krisen- und Ereignisfall-Management
- Abteilung Landesinstitut für Lebensmittel, Lebensmittelhygiene und kosmetische Mittel, Sachbereich Lebensmittelhygiene Süd
- Abteilung Landesinstitut für Gesundheit (Sachgebiet für Hygiene / Sachbereich Infektionsepidemiologie / Sachbereich Bakteriologie und Mykologie )
- Landratsamt Weilheim-Schongau, Veterinäramt / Amt für Verbraucherschutz
Beneficiary: Dr. Christiane Wagner-Wiening
Institution: State Health Authority of Baden-Württemberg
Funding period: 1.1.2019 - 31.12.2020
Brief description
Since 2014 the occurrence of local Aedes albopictus populations has been observed in Baden-Württemberg. How can the public health service at the municipal level be prepared for the prevention and control of vector-competent mosquitoes and arboviroses through the transfer of scientific knowledge? Which legal foundations have to be considered? Which communal responsibilities and communication channels apply to the prevention and control of Ae.albopictus ? Which decision guidance can support the ÖGD in the prevention and control of Ae.albopictus?
The main objective of the project is to establish a stakeholder network that carries out monitoring and intervention structures for vector-competent mosquitoes and possible autochthonous arbovirus infections at the municipal level. The main focus of the project is a network analysis as well as the consolidation and harmonisation of already existing research results and recommendations for action. Stakeholders and their functions in the implementation of control and eradication measures will be identified by means of expert interviews, and research results and experiences on prevention and intervention measures will be reviewed and evaluated by means of literature research and expert interviews. Within the scope of the stakeholder analysis, interests and framework conditions are to be taken into account, measures derived and communication channels described.
On the basis of the results obtained, an arbovirus and mosquito management plan will be developed as a guideline for the municipal level and validated by means of focus group discussions. The plan will define the legal basis, responsibilities and decision-making aids for the prevention and control of vector-competent mosquitoes and mosquito-borne arboviroses.
Definition of terms:
Arbovirus: Arbo = acronym for arthropod-borne viruses, an arbovirus is a virus transmitted by arthorpods (= arthropods, such as mosquitoes, e.g. Aedes albopictus). A well-known representative is the TBE virus (TBE = early summer meningo-encephalitis).
Arbovirosis: Disease caused by an arbovirus infection
Beneficiary: Dr. Jasmin Skuballa
Chemical and Veterinary Investigation Office Karlsruhe
Funding period: 1.3.2019 - 28.2.2021
Background:
Was viele Verbraucher nicht wissen: die zoonotische Infektionskrankheit Frühsommer-Meningoenzephalitis (FSME) kann neben einer Übertragung via Zeckenstich auch über virushaltige Rohmilch von Schafen, Ziegen und Rindern zur Erkrankung bei nicht geimpften Menschen führen.
Die serologische Untersuchung von Wiederkäuern mit Weidehaltung eignet sich hervorragend, kleinräumige FSME-Naturherde aufzuspüren, da diese Tiere nach Infektion über Monate oder Jahre hinweg einen detektierbaren Antikörper-Titer ausbilden („Sentinels“).
Objectives:
Das Ziel der Studie ist die Datenerhebung zur Risikoabschätzung alimentärer FSME-Infektionen durch Untersuchung von Wiederkäuern als Sentinel-Tiere. Die im Projekt gewonnenen Daten können helfen, das Infektionsrisiko für den Menschen durch den Verzehr roher Milchprodukte besser abschätzen zu können und somit zu einem besseren Verbraucherschutz beitragen.
Scheduled implementation:
1st project phase:
Serological screening of blood samples from ruminants will be used to identify TBE natural foci in Baden-Württemberg.
2nd project phase:
After the identification of a natural TBE herd, the targeted serological and molecular biological examination (ELISA, real-time PCR) of blood and milk samples from a cooperative dairy farm over a period of one year follows.
3rd project phase:
Investigation of host-seeking ticks in the vicinity of the livestock pasture to determine a possible correlation between findings in the investigated animals and human disease cases in the region.
4th project phase:
The data will be evaluated and a risk analysis for the affected region will be made.
Cooperation:
State Health Office (LGA) Stuttgart, Mr. Dr. Oehme
(cultivation of TBE isolates in the BSL3 laboratory, phylogenetic studies)
Institute for Microbiology of the German Armed Forces (IMB), Munich
Consiliary Laboratory for Early Summer Meningoencephalitis, Mr. Dr. Dobler
(further testing of positive serum samples, phylogenetic studies)
Beneficiary: Dr. Stefan Brockmann
Institution: District of Reutlingen
Funding period: 1.12.2018 - 30.11.2021
Brief description
The ENV-HANTA project aims at the detection of hantaviruses from environmental samples in the immediate vicinity of households of formerly ill persons in high-endemic areas for hantaviruses. The results will be used to make existing recommendations for protective measures more specific.
The project promotes application-oriented research on the prevention and control of zoonotic infectious diseases. Eight local public health authorities in Baden-Württemberg are cooperating with the State Health Office, the University of Tübingen, a faunistic expert office and the Friedrich-Loeffler Institute. The project makes a concrete contribution to the control of rodent-associated diseases and follows the "One Health" approach.
The results of the project should make it possible to gain knowledge about the actual occurrence of hantaviruses in/around households in endemic areas as a possible source of infection.
With the results it is possible to specify the recommendations of the public health service to the population, e.g. which protective measures should be implemented for which activities. Thus a reduction of the disease burden can be achieved.
The project is funded for three years (start: December 2018).
Background
Puumala Hantavirus (PUUV) (family Bunyaviridae) is a zoonosis with increasing importance in public health in Europe. In Germany, hantaviruses are among the priority pathogens for surveillance and epidemiological research. PUUV occurs in bank voles; an increased population density of these rodents in certain years is associated with human hantavirus epidemics. Reddish mice excrete the viruses with their excretions. Transmission to humans occurs mainly by inhalation of virus-containing aerosols. In humans, the infection can result in nephrological hantavirus disease. Recent studies suggest that this disease can lead to chronic secondary symptoms. In Germany, more than 50% of human PUUV cases are reported from Baden-Württemberg. In the last 17 years, three major epidemics of more than 1,000 cases each have been reported.
In order to prevent human hanta virus diseases as efficiently as possible, it must be known where and during which activities people become infected. This point is to be clarified in the present project. In particular, evidence from case-control studies will be checked by pathogen detection from environmental samples in order to identify the stay at certain places or activities carried out there that are relevant for transmission.
A literature search in a scientific online citation and literature database (Web of Science) showed that there are hardly any scientific studies dealing with the survivability of hantaviruses in the environment. Studies on findings on the presence of PUUV at possible infection sites do not yet exist. In its "Technical Report - Prevention measures and communication strategies for hantavirus infection in Europe", the European Centre for Disease Prevention and Control (ECDC) stated that the prevention measures applied in 29 EU member states to date are hardly evidence-based and their impact has not yet been sufficiently evaluated. The ENV-HANTA project could contribute an important building block to the clarification of these questions by the detection in environmental samples at defined places in the domestic environment.
To complement the results of the environmental samples, additional rodents will be trapped at the test sites and tested for hantavirus. The findings from the project may make it possible to specify intervention measures that are generally recommended (e.g. the wearing of protective masks) more precisely and use them in a more targeted manner.
Extract from the literature list of applicants for hantavirus diseases
- S.O. Brockmann, M Schwehm, C Wagner-Wiening, G Kändler, M Eichner. Forecasting the incidence of human Puumala virus cases in South West Germany.2015. European Scientific Conference in Applied Infectious Diseases Epidemiology( ESCAIDE) Nov 7th Stockholm, Sweden
- Latus J, Schwab M, Tacconelli E, Pieper FM, Wegener D, Dippon J, Müller S, Zakim D, Segerer S, Kitterer D, Priwitzer M, Mezger B, Walter-Frank B, Corea A, Wiedenmann A, Brockmann S, Pöhlmann C, Alscher MD, Braun N. Clinical course and long-term outcome of hantavirus-associated nephropathia epidemica, Germany. Emerg Infect Dis. 2015 Jan;21(1):76-83. doi: 10.3201/eid2101.140861.
- Latus J, Schwab M, Tacconelli E, Pieper FM, Wegener D, Rettenmaier B, Schwab A, Hoffmann L, Dippon J, Müller S, Fritz P, Zakim D, Segerer S, Kitterer D, Kimmel M, Gußmann K, Priwitzer M, Mezger B, Walter-Frank B, Corea A, Wiedenmann A, Brockmann S, Pöhlmann C, Alscher MD, Braun N. Acute kidney injury and tools for risk-stratification in 456 patients with hantavirus-induced nephropathia epidemica. Nephrol Dial Transplant. 2015 Feb;30(2):245-51. doi: 10.1093/ndt/gfu319. Epub 2014 Oct 13.
- Faber MS, Ulrich RG, Frank C, Brockmann SO, Pfaff GM, Jacob J, Krüger DH, Stark K. Steep rise in notified hantavirus infections in Germany, April 2010. Euro Surveill. 2010 May 20;15(20). pii: 19574. PubMed PMID: 20504391.
- Schwarz AC, Ranft U, Piechotowski I, Childs JE, Brockmann SO. Risk factors for human infection with Puumala virus, southwestern Germany. Emerg Infect Dis. 2009 Jul;15(7):1032-9. doi: 10.3201/eid1507.081413. PubMed PMID: 19624917; PubMed Central PMCID: PMC2744254.
- Winter CH, Brockmann SO, Piechotowski I, Alpers K, an der Heiden M, Koch J, Stark K, Pfaff G. Survey and case-control study during epidemics of Puumala virus infection. Epidemiol Infect. 2009 Oct;137(10):1479-85. doi: 10.1017/S0950268809002271. Epub 2009 Mar 17. PubMed PMID: 19288958.
- Piechotowski I, Brockmann SO, Schwarz C, Winter CH, Ranft U, Pfaff G. Emergence of hantavirus in South Germany: rodents, climate and human infections. Parasitol Res. 2008 Dec;103 Suppl 1:S131-7. doi: 10.1007/s00436-008-1055-8. Epub 2008 Nov 23. PubMed PMID: 19030895.
- Koch J, Brockmann SO, Winter C, Kimmig P, Stark K. Significant increase of hantavirus infections in Germany since the beginning of 2007. Euro Surveill. 2007 May 3;12(5):E070503.1. PubMed PMID: 17868605.
- Kimmig P, Silva-González R, Backe H, Brockmann S, Oehme R, Ernst E, Mackenstedt U. Epidemiology of hantaviruses in Baden-Wurttemberg. Gesundheitswesen. 2001 Feb;63(2):107-12. German. PubMed PMID: 11285748.
Beneficiary: Dr Jobst Heitzig
Institution: Potsdam Institute for Climate Impact Research
Funding period: 1.03.2019 - 28.02.2022
Project description
Links between livestock, for example through the movement of animals to other herds (livestock trade), play a major role in the spread of animal diseases. In the project, information on the structure of this network will be used to design optimized designs of sample surveys. Which investigation effort can be used to achieve the greatest benefit in order to demonstrate freedom from an animal disease / zoonosis or to detect an infection that has occurred? The theoretical literature knows a number of disease propagation models on networks and provides basic insights into which network nodes are crucial for propagation. These findings can help to develop test heuristics (e.g. test networks with a higher number of connections in the network more frequently). Based on test heuristics that seem promising in light of the literature, we want to use current methods of optimization and machine learning to determine optimal test designs for specific epidemics and regions. It is possible to estimate the change in the significance of favourable results of key-date surveys with regard to the health status (one herd or all herds in the area are free of infection) with a time lag from the day of the survey, if the cross-linking of the animal populations and the probability of an intermediate introduction of the infectious agent are taken into account. In this way, test designs for granting the status "free from infection" can be optimised in a risk-oriented way.
Some infections initially proceed clinically inconspicuously over a longer period of time. The health status of an animal population is usually derived from studies in animal populations. This can be done by examining each animal in the herds or all herds or by random sampling. Since the examinations are not carried out daily, the question arises as to what significance such examinations still have at a later date and how this changes over time. This question arises for each individual herd and also for the herds in an area for which a health status is granted. In the project this will be analysed and presented using mathematical and epidemiological methods, taking into account the knowledge of infection dynamics. In addition to the connectivity of the herds (contact possibilities/introduction possibilities), the distribution of the respective infectious agent is of great importance for the probability of an intermediate pathogen entry into the animal population; the general distribution depends for example on climate change. One example is the spread of the West Nile Fever virus, whose vectors (certain species of mosquito) are increasingly drifting northwards as a result of climate change.
The issue is of global importance, particularly in international trade in animals and animal products; the more reliable the health status granted, the better animal populations and humans are protected against the introduction of an infection. The knowledge gained in this project will be made available to the scientific community and practitioners through scientific publications and stakeholder dialogues and will help to implement the risk-based approaches required by the planned EU legislation on the reform of animal health law.
Cooperation partner
Dr. Jörg Fritzemeier, Veterinary Service for City and County of Osnabrück
Funding recipient: Prof. Dr. Lothar Kreienbrock
Institution: University of Veterinary Medicine Hannover Foundation
Funding period: 1.4.2019 - 31.03.2022
Cooperation partners
-
Dr. Jörg Fritzemeier, Veterinary service for city and district of Osnabrück
-
Prof. Dr. Eberhard Haunhorst, Lower Saxony State Office for Consumer Protection and Food Safety
Project description
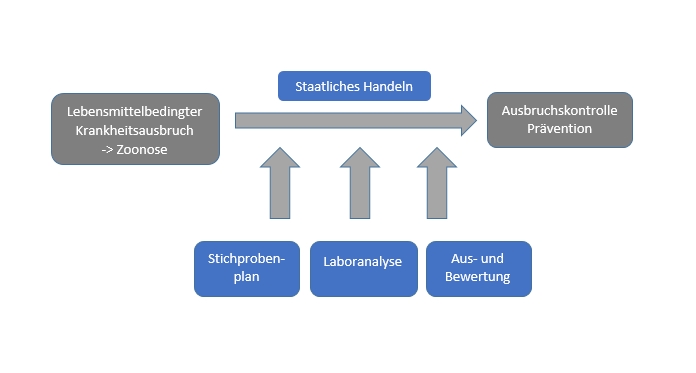
For specific questions of food law, it is always necessary to develop and apply appropriate sampling plans. This need for sampling arises, inter alia, in cases of food-borne zoonotic disease outbreaks, as well as in the course of assessing whether food batches or food production processes are considered safe with regard to their zoonotic burden. The method of sampling and detection chosen shall be representative and meaningful in order to provide a sufficient degree of confidence in the presence of the zoonotic agent. The selection of appropriate methods regularly poses challenges to the competent authorities in this respect. Often complex interrelationships have to be considered, such as, inter alia, the characteristics of the pathogen, the timing and type of introduction, the homogeneity of distribution, the matrix and the laboratory method to be used. The type of sampling plan must also be adapted from one project to another.
Various tools are currently available to help answer such questions. Frequently used are the tools for calculating sample sizes of the European Food Safety Authority (EFSA) or the Predictive Microbial Modeling Lab (PMM-Lab) of the Federal Institute for Risk Assessment for modeling bacterial growth or the inactivation of bacteria and toxins. However, both tools only offer a part of the required functionality to make a final statement. In addition, it is often necessary to specify unknown variables, such as the prevalence of the analyte in the matrix to be examined.
The StiproZoo project aims to answer the research question of whether it is possible to develop a meaningful, feasible and at the same time biometrically correct sampling scenario taking into account a wide range of factors such as infectiology, production technology and logistics. Due to the complexity of the issue, selected practical scenarios are to be given priority and a generic concept for all pathogens is to be developed.
The long-term goal is to develop an application-oriented sampling tool (Shiny-App) by combining scientific knowledge about the pathogen, technological expertise in food production and epidemiological-scientific knowledge. The application should illustrate in a modular way the different levels to be considered in sample design and analysis. This work aid and the associated findings are to be made available to the responsible authorities.
Beneficiary: Dr. Cornelia Henke-Gendo
Institution: Lower Saxony State Health Authority
Funding period: 1.3.2019 - 28.02.2022
Summary:
Animals are able to address and influence people in a special way. In certain contexts, interaction with animals can even have healing effects. In addition, assistance dogs, for example, act as important supporters for people with special needs (e.g. guide dogs for the blind) or even as their lifesavers (warning dogs for diabetics). It is therefore not without reason that offers in the field of animal-supported therapies and other animal-supported interventions are also constantly increasing in medical and care facilities. In addition to all these positive aspects, however, it should also be noted that interaction with animals can lead to the transmission of zoonotic pathogens. This fact takes on a special significance especially in medical institutions when it comes to contact with people with deficiencies in immune defence (e.g. sick people, children, elderly people). Up to now, there are no uniform guidelines for the field of animal-supported interventions, with which measures a potential zoonotic risk can be counteracted in order to minimize it. Various national and international recommendations differ, sometimes considerably, and the evidence behind these recommendations is not always clear.
In the project METT (Multiresistant pathogens in animal-assisted therapy) we want to develop a data basis that allows a risk analysis for the transmission of zoonotic pathogens in the context of animal-assisted therapies in medical institutions. A special focus will be on multi-resistant pathogens relevant for hospital hygiene, such as MRSA and multi-resistant Gram-negative bacteria. In a first step, the extent and type of human-animal contacts in medical facilities and other health and, if applicable, welfare institutions in Lower Saxony will be recorded and existing guidelines for dealing with a potential risk of infection evaluated. The owners of the animals used in this process are offered the opportunity to have their animals examined for the presence of multi-resistant pathogens. The evaluation of the collected data is to result in recommendations for action for the individual facilities and, if necessary, also for the animal owners and create the basis for an appropriate and comparable infection-hygienic monitoring of this area by the health authorities. For this purpose, hygienically relevant quality markers will be defined in cooperation with representatives of the health authorities of Lower Saxony and made available in the form of an inspection checklist. The state-wide use of such a checklist for inspection and reflection of the monitoring results to the Lower Saxony State Health Authorities will allow, after evaluation of the data, an assessment of the frequency and quality of human-to-animal contacts and thus also an estimation of the risk for the transmission of pathogens from animals to humans.




